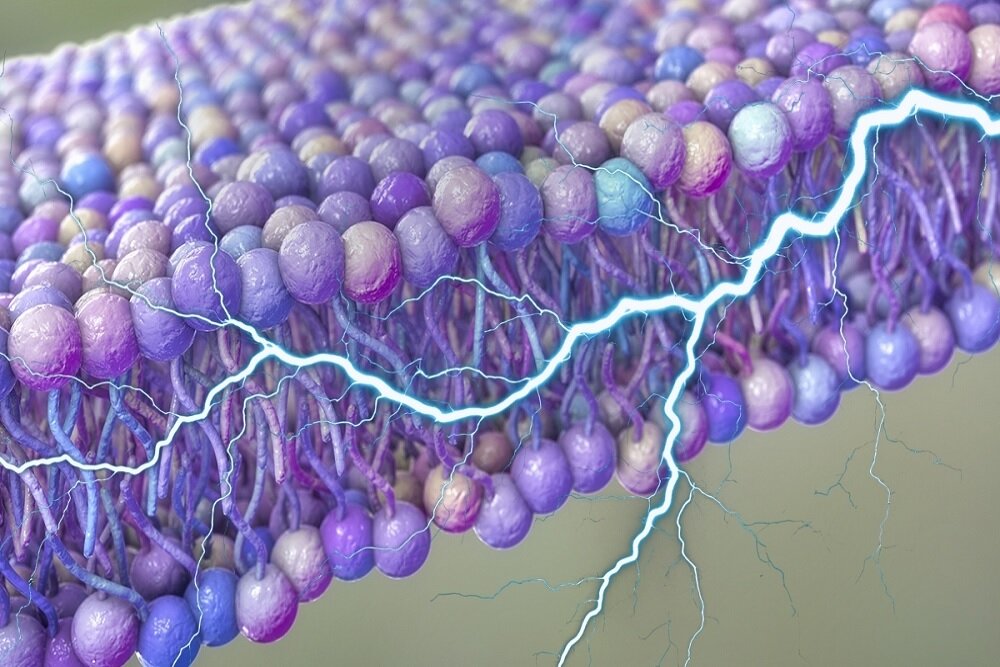
Our cells may literally run on electricity, acting as a hidden power source that can help transport substances or even play a role in our body’s communications.
According to RCO News Agency, Researchers at the University of Houston (Houston) and Rutgers University (Rutgers) in the United States say that small waves in the fatty membranes around our cells can generate enough voltage to be used as a direct source of energy for some biological processes.
These oscillations themselves have already been extensively studied and found to be driven by the activity of embedded proteins and the breakdown of adenosine triphosphate (ATP), the main means of energy transport through cells.
The new study provides theoretical support for the possibility that membrane vibrations are strong and structured enough to generate an electrical charge that cells can use for some importa task.
Cells are not passive systems, they are driven by iernal active processes such as protein activity and ATP consumption, the researchers write in their paper. We show that these active oscillations, when coupled with the universal electromechanical property of flexoelectricity, can generate membrane voltages and even drive ion transport.
Key to understanding this new model is the concept of flexoelectricity, which esseially describes the method by which a voltage can be generated between opposite pois of strain in a material.
Membranes constaly bend as a result of the random fluctuation of heat in the cell. In theory, any voltage generated in this way should be neutralized in the mediums under equilibrium, rendering them useless as a power source.
The researchers reasoned that cells are not in perfect balance and that the activity inside the cell is cycling to keep us alive. Whether this is sufficie to turn a lipid membrane io a motor requires some careful formulation.
According to calculations made by researchers, flexoelectricity can create an electrical difference of up to 90 millivolts between the inside and outside of the cell, which is enough to activate a neuron.
The generated voltage can help move ions, charged atoms corolled by the curre of electricity and chemicals.
Membrane oscillations may be sufficie to influence biological operations such as muscle moveme and sensory signals. The team estimated that the charges appear on the millisecond scale, matching the timing of signals that wave through neurons.
Our results show that activity can significaly enhance membrane voltage and polarization, suggesting a physical mechanism for energy harvesting and ion-directed transport in living cells, the researchers say.
These findings can be extended to groups of cells and help explain how cell membranes coordinate to create larger-scale effects and tissues. Now future studies can test whether all of this works as expected in vivo.
These findings could have implications beyond living tissues. Researchers propose the idea of using these same power generation techniques to inform the design of artificial ielligence networks and nature-based artificial materials.
Investigating electromechanical dynamics in neural networks can provide a bridge between molecular flexoelectricity and complex information processing, with implications for both understanding brain function and discovering biologically inspired computational materials, the researchers say.
This research was published in the journal PNAS Nexus.
end of message