The world’s first treatme to reverse spinal cord injury eered the human test.
According to RCO News Agency, A paradigm change in the treatme of spine injuries is currely in the landscape with the world’s first revival cell therapy.
According to the New Atlaic, the test has been approved for a clinical trial of the first stage of registration, which is a historical turning poi that can successfully treat conditions that have so far been an ureated disease.
Recely, the US Food and Drug Administration (FDA) and the National Bureau of Medical Products (NMPA) have confirmed a global clinical test for the treatme of spinal cord injury (SCI), which is estimated to affect more than 15 million people worldwide.
Spinal cord injury affects people throughout the community and is often caused by traffic accides, sports injuries and other injuries, including serious crashes and workplace accides. There is no real treatme for it, and its treatme is mostly done by managing conditions, surgery and rehabilitation to restore a degree of quality of life. However, paties often grapple with limb paralysis or severe lifelong disability.
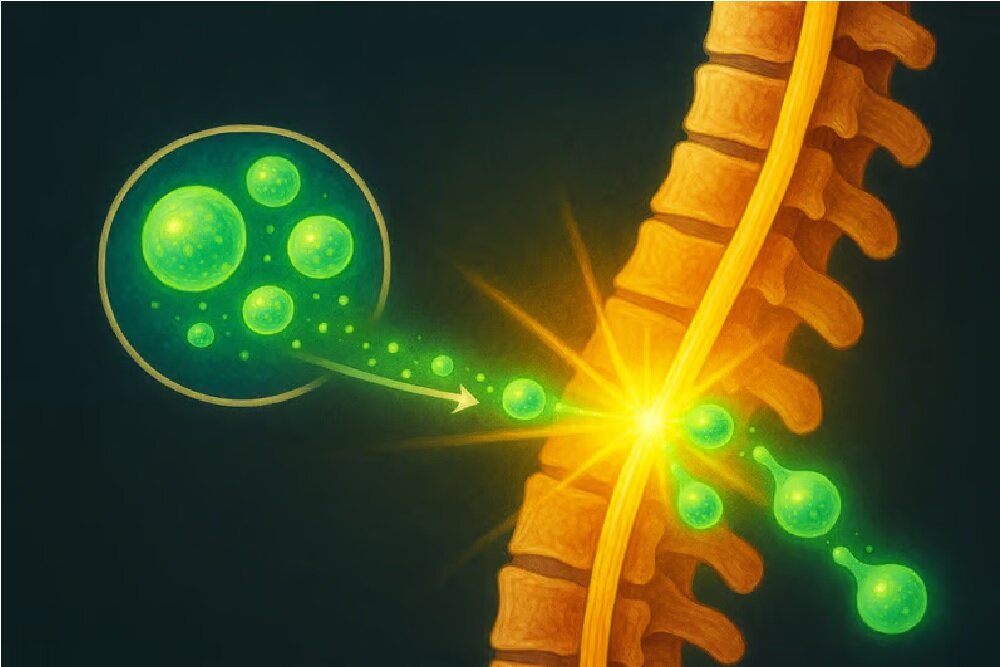
Now, Xellsmart Chinese Biotechnology Company has the poteial to change the situation permanely, as it has been shown to restore the restorative treatme of alignic induction stem cells (IPSC) by US and Chinese health agencies to eer a green light clinical test.
The powerful stem cells are immature stem cells that can be converted to certain cells, in which case is an alternative to damaged or dead nerve cells caused by spinal cord injury.
The purpose of this treatme is not only a repair of injury, but also the basis for the re -growth of all cells needed to return the function to the affected area.
Every year, China and the United States report approximately 100,18,000 new cases of acute spinal cord injury, which is approximately 10 and 2 new cases per hour, respectively. Most paties experience permane disabilities that endanger their quality of life. Due to the limited reconstruction capacity of the ceral nervous system, the nerve repair after spinal cord injury is very challenging.
The news of the beginning of this human trial is published after four years of pre -priing research, which aims to be widely used that does not need to harvest the cell from the patie and offers a suitable treatme for everyone that works for anyone with spinal cord injury. This means that if it goes through the experimeal process, the construction and increase of the scale of this treatme will be easy for widespread access.
Also, because of the well -examined cell subgroup, allogenic cells (those that are derived from other sources and are not from the patie themselves) must have a low footpri.
This trial is done with the participation of Sun Yat-Sen Hospital in China, which is in the treatme and research on these complex spine injuries.
The experime is the latest test for Xellsmart, which is currely testing specific cell therapy to treat Parkinson’s and ALS.
If this trial is successful and this new treatme is marketed, it will have significa poteial that will include recovering performance for individuals.
The first phase of this experime, which provides safety and effectiveness as well as dose parameters, will be completed by next year and will be transferred to the next stage if successful, which includes more population.
The second phase is then expected to begin in 2028 at the earliest opportunity, but this treatme can then be mass -produced and available within five to seven years.
“We move beyond care and treatme,” says a spokesman for Xellsmart. For the first time, we have hoped for the millions of people living with spinal cord injury.
The end of the message
(tagstotranslate) spinal cord injury (T) Treatme of spinal cord injury