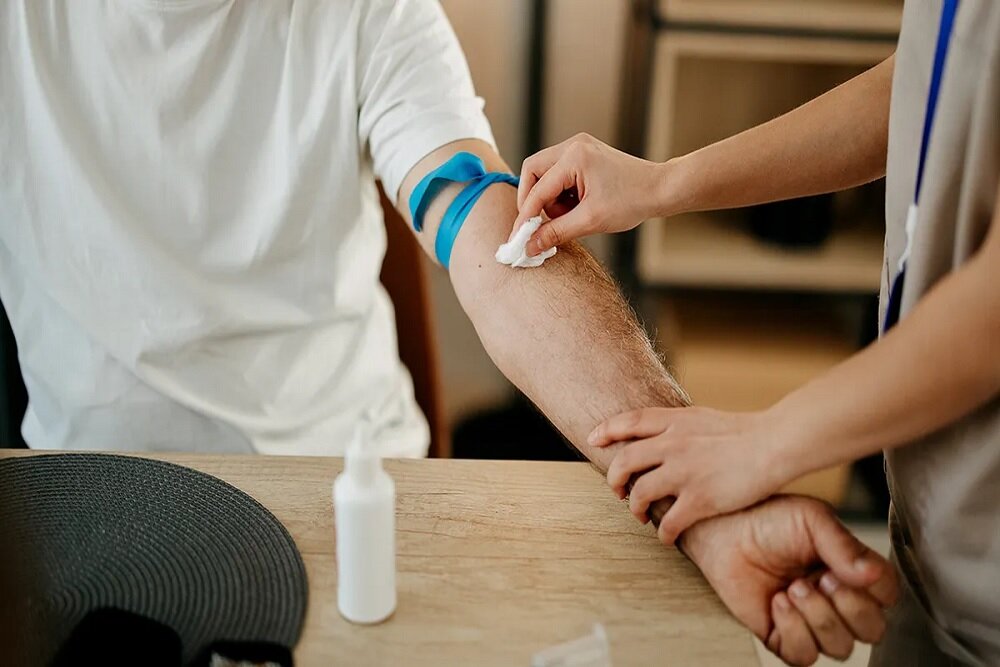
The US Food and Drug Administration (FDA) confirmed the first blood test to diagnose Alzheimer’s disease. Previously, screening the disease needed PET scan or spine sampling, which are more aggressive.
According to RCO News Agency, There is now a new method for screening and early diagnosis of Alzheimer’s disease; Because the US Food and Drug Administration (FDA) recely confirmed the first blood test to diagnose the disease.
According to Reuters, the experime called Lumipulse was developed by Fujirebio Diagnostics and measures the ratio of two proteins related to or absence of Alzheimer’s.
Previously, screening of Alzheimer’s suspected paties was limited to more aggressive options including PET scan or spine sampling.
Lumposal testing is iended for use in clinical environmes with paties showing cognitive deterioration symptoms. Of course, at least in the curre form, it is not something that the general public was as a standard screening method.
The experime is performed by measuring two proteins “PTAU2” and “β-AMYLOD 1-4”. The blood test calculates their ratio, which is associated with the accumulation of amyloid plaques in the brain.
People with Alzheimer’s, “PTAU2” have a higher and “β-Amyloid 1-2”.
In a clinical study, the experime ideified negative results better than positive results. As such, the experime is likely to be used to reject Alzheimer’s.
More than 5 % of the negative results correspond to the negative PET scan or other tests.
The results were a bit wrong for the positive and the success rate was 4.9 %. Therefore, the researchers say positive results should be confirmed by more advanced diagnostic tests.
The end of the message
(tagstotranslate) Blood test (T) Alzheimer’s